Avoiding Pump Cavitation in Open Systems: Why Pump Suction Pressure and Water Temperature are Critical
/By Chad Edmondson
How can you be certain that the pump you select will not cavitate in the system you are designing? By now it should be clear that such certainty is a matter of analyzing the following variables:
• Available Suction Flange Pressure – What will the pressure gauge reading be at the suction of the pump?
• The required NPSH for the pump you are considering. Remember, this information is available from the pump manufacturer and is typically found at the bottom of the pump curve. A pump’s required NPSH is equivalent to the amount of pressure drop between the pump suction and the eye of the impeller.
• The temperature of the water you are pumping.
• The boiling point of water for your particular elevation. The higher your elevation the lower the boiling point and the easier it is for the pump to cavitate.
So let’s go over some simple examples, based on the assumption that our system is at sea level, in which case the boiling point for water is 212°F at 0 psig. Why is that important to know? Because it tells us that we cannot let water drop below 0 psig if there is any chance its temperature could reach 212°F.
Example 1
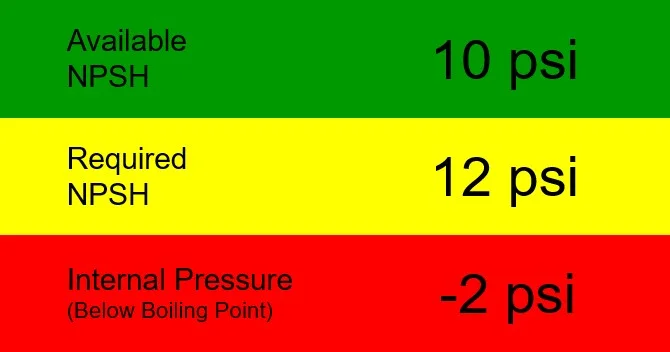
We are designing a system that will be required to pump 212°F water from an open tank located above the pump. We have measured the vertical height from the surface of a hot water tank and know that it is 23.1 feet above the pump suction. That translates into 10 psi of pressure at the suction of our pump. In other words, that is the reading we will get on the pressure gauge at the pump suction. Our available NPSH is 10 psig (vertical static height) minus 0 psig (vapor pressure of water at 212°F). Finally, we are looking at pump with a required NPSH of 4 psi.
What will be the internal pressure of this pump? The answer is 6 psi, because we must subtract the required NPSH from the available NPSH. That leaves us 6 psi, a positive pressure that will keep the water from boiling in the pump.
Thumbs up on the pump selection. We won’t cavitate!
Example 2
Now let’s say we want to trim cost, so we consider a smaller pump. It has a higher required NPSH (NPSHR) because, all other things being equal, smaller pumps always have higher NPSHR for a given operating point than larger pumps. In this case, our required NPSH is 12 psi. That leaves us with a negative internal pressure of -2 psi. That’s trouble because we know we have to remain above 0 psi to eliminate any chance for cavitation. If our water is 212°F, cavitation is inevitable. Better look for another pump – or replace the one that was misapplied on the existing system.
Keep in mind that when calculating available NPSH the lower the water temperature then the lower the vapor pressure of water. For cooling tower applications, the vapor pressure of water at 85°F is -14 psi. If we were pumping 85°F water instead of 212°F in the example above, we would have an available NPSH of 24 PSI! Because of the role that temperature plays in available NPSH, most NPSH problems are likely to occur in steam condensate systems or open systems where the pump is located above a tank. This also serves as a good reminder to always specify compound pressure gauges for open systems pumps. You need to be able to confirm the exact pressure on the suction of a pump, positive or negative.