Cooling Tower and Condenser Water Design Part 9: Controlling Cycles of Concentration
/By Chad Edmondson
Successful cooling tower operation and performance involves a delicate balance between (1) keeping the dissolved solids (such as calcium, magnesium, chloride, and silica) in the water from causing too much scale and/or corrosion and (2) conserving make-up water. It’s expensive, inefficient, and environmentally irresponsible to continuously dilute cooling tower water with an abundance of make-up water just to keep the ratio of minerals down. But how do you decide how much make-up water is needed to keep minerals within a tolerable range?
Understanding Cycles of Concentration
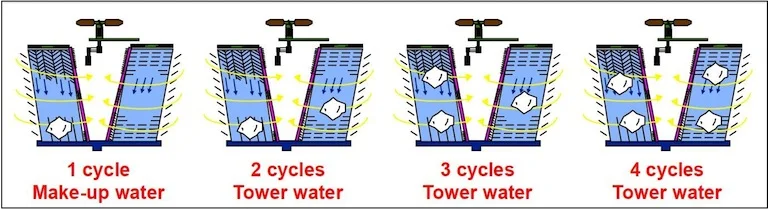
The key is controlling the cooling tower’s cycles of concentration. The terminology, “cycles of concentration”, is merely a comparison between the level of solids of the recirculating cooling tower versus the level of solids of the original raw make-up water. Thus, if the circulating water has four times the solids concentration than the make-up water, then the cycles of concentration are 4.
For the record, most cooling towers will operate fine at up to 4 or 6 cycles of concentration, meaning that they can successfully operate with up to 4 or 6 times the amount of dissolved solids that was in the original make-up water. This, however, will depend on a few other variables, including the quality of the water in your area, and the chemical treatment plan you have chosen.
Conserving Cooling Tower Water
Engineers or plant managers must specify what the desired cycles of concentration will be and will control the cooling tower blow-down (bleed off) accordingly. Blow-down or bleed off is the water that is drained from the cooling equipment in order to remove excessive mineral build-up. Bleed off too much and you waste water; bleed off too little and you get scale and/or corrosion.
It is advisable that the amount of bleed off be automatically controlled by a system that actually measures the amount of solids concentration in the water – although that’s not always the case. Manual bleed-offs are very common, but most certainly on their way out as concerns over water shortages continue to grow throughout the US. To some extent, building codes already regulate the water consumption of cooling towers, and that regulation continues to increase. Standard 189.1 – 2009 for the Design of High Performance Green Buildings, a standard which the Army Corp of Engineers has already adopted as have many other new construction buildings, include the following water conservation requirements for cooling towers:
- Cooling towers must be equipped with make-up and blow-down meters, conductivity controllers, and overflow alarms in accordance with specific thresholds listed in the standard.
- Cooling towers must also be equipped with efficient drift eliminators that reduce drift to a maximum of 0.002% of the recirculated water volume for counter-flow towers and 0.005% for cross-flow towers.
- Water discharged from a cooling tower used for air conditioning must be limited depending on the water hardness. A minimum of five cycles of concentration is required for make-up water having less than 200 ppm of total hardness (calcium carbonation). A minimum of 3.5 cycles of concentration is required for make-up water with more than 200 ppm of total hardness. The only exception to this is water which exceeds 1500 mg of dissolved solids or 150 pp of silica.
Also note there are additional mandatory provisions in this standard that address water efficiency elsewhere in the cooling system. One such provision worth mentioning here is the requirement that condensate from air conditioning units with a capacity greater than 65,000 Bth/h (19kW) be recovered for re-use. You might note that this isn’t a particularly large cooling system – a mere 5 tons. Air handler condensate is a good option for recovering for cooling tower make-up water since the condensate has a low mineral content and is typically the most plentiful when cooling tower loads are the highest.
For more information, please view our video series on Cooling tower and Condenser Water Piping Design.